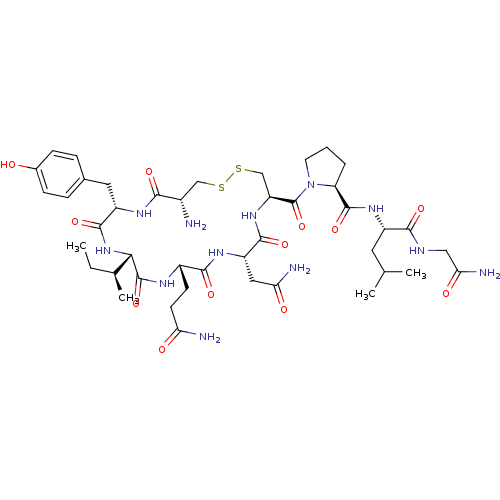
 BDBM50205990 CHEMBL395429 OXYTOCIN
BDBM50205990 CHEMBL395429 OXYTOCIN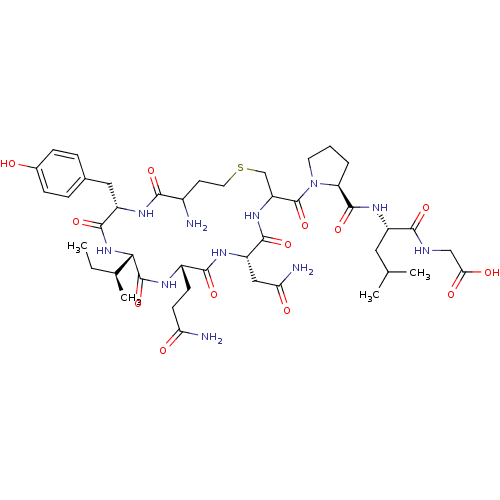 BDBM50332723 Cystathionine Oxytocin CHEMBL1630531
BDBM50332723 Cystathionine Oxytocin CHEMBL1630531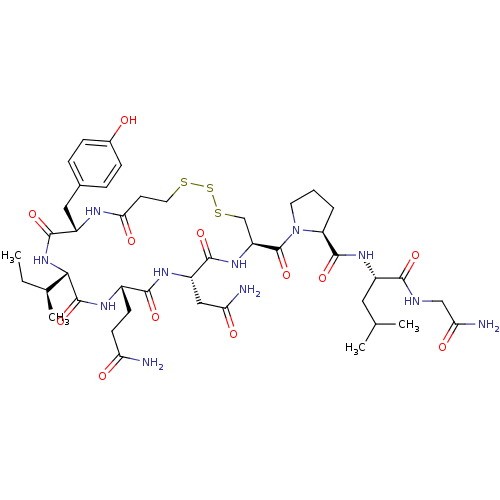 CHEMBL352836 BDBM50056797 Oxytocin Trisulfide
CHEMBL352836 BDBM50056797 Oxytocin Trisulfide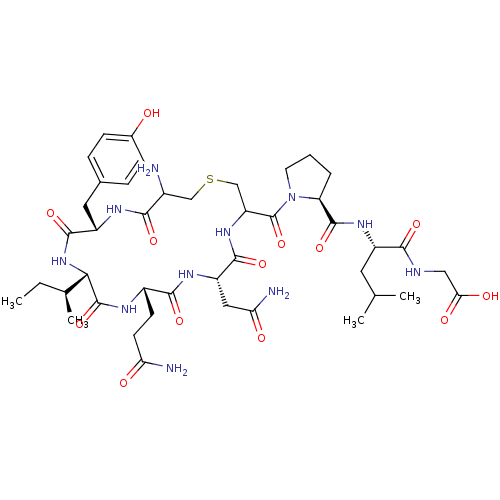 Lanthionine Oxytocin BDBM50332726 CHEMBL1630530
Lanthionine Oxytocin BDBM50332726 CHEMBL1630530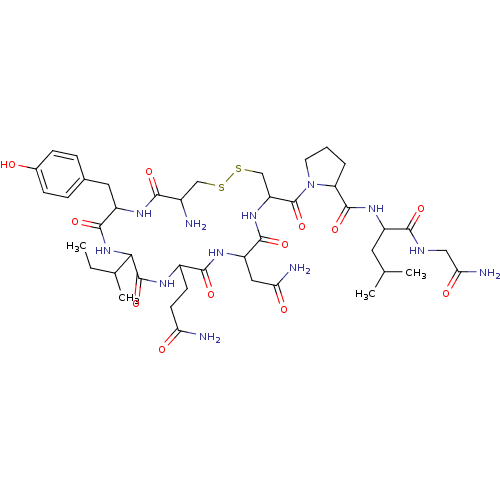 CAS_50-56-6 Oxytocin BDBM86210 NSC_439302
CAS_50-56-6 Oxytocin BDBM86210 NSC_439302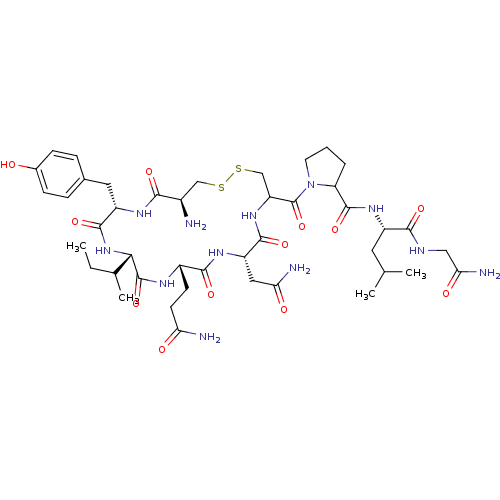 Cys-Tyr-Ile-Gln-Asn-Cys-Pro-Leu-Gly-NH2 1-[19-Amino-13-sec-butyl-10-(2-carbamoyl-ethyl)-7-carbamoylmethyl-16-(4-hydroxy-benzyl)-6,9,12,15,18-pentaoxo-1,2-dithia-5,8,11,14,17-pentaaza-cycloicosane-4-carbonyl]-pyrrolidine-2-carboxylic acid [1-(carbamoylmethyl-carbamoyl)-3-methyl-butyl]-amide CHEMBL265640 1-[19-Amino-13-sec-butyl-10-(2-carbamoyl-ethyl)-7-carbamoylmethyl-16-(4-hydroxy-benzyl)-6,9,12,15,18-pentaoxo-1,2-dithia-5,8,11,14,17-pentaaza-cycloicosane-4-carbonyl]-pyrrolidine-2-carboxylic acid [1-(carbamoylmethyl-carbamoyl)-3-methyl-butyl]-amide(Oxytocin) 1-[19-Amino-13-sec-butyl-10-(2-carbamoyl-ethyl)-7-carbamoylmethyl-16-(4-hydroxy-benzyl)-6,9,12,15,18-pentaoxo-1,2-dithia-5,8,11,14,17-pentaaza-cycloicosane-4-carbonyl]-pyrrolidine-2-carboxylic acid [1-(carbamoylmethyl-carbamoyl)-3-methyl-butyl]-amidec-[Cys-Tyr-Ile-Gln-Asn-Cys]-Pro-Leu-Gly-NH2 BDBM50013775 OXYTOCIN (oxytocin-OT) cyclo[Cys-Tyr-Ile-Gln-Asn-Cys]-Pro-Leu-Gly-NH2
Cys-Tyr-Ile-Gln-Asn-Cys-Pro-Leu-Gly-NH2 1-[19-Amino-13-sec-butyl-10-(2-carbamoyl-ethyl)-7-carbamoylmethyl-16-(4-hydroxy-benzyl)-6,9,12,15,18-pentaoxo-1,2-dithia-5,8,11,14,17-pentaaza-cycloicosane-4-carbonyl]-pyrrolidine-2-carboxylic acid [1-(carbamoylmethyl-carbamoyl)-3-methyl-butyl]-amide CHEMBL265640 1-[19-Amino-13-sec-butyl-10-(2-carbamoyl-ethyl)-7-carbamoylmethyl-16-(4-hydroxy-benzyl)-6,9,12,15,18-pentaoxo-1,2-dithia-5,8,11,14,17-pentaaza-cycloicosane-4-carbonyl]-pyrrolidine-2-carboxylic acid [1-(carbamoylmethyl-carbamoyl)-3-methyl-butyl]-amide(Oxytocin) 1-[19-Amino-13-sec-butyl-10-(2-carbamoyl-ethyl)-7-carbamoylmethyl-16-(4-hydroxy-benzyl)-6,9,12,15,18-pentaoxo-1,2-dithia-5,8,11,14,17-pentaaza-cycloicosane-4-carbonyl]-pyrrolidine-2-carboxylic acid [1-(carbamoylmethyl-carbamoyl)-3-methyl-butyl]-amidec-[Cys-Tyr-Ile-Gln-Asn-Cys]-Pro-Leu-Gly-NH2 BDBM50013775 OXYTOCIN (oxytocin-OT) cyclo[Cys-Tyr-Ile-Gln-Asn-Cys]-Pro-Leu-Gly-NH2
- Borthwick, AD Oral oxytocin antagonists. J Med Chem 53: 6525-38 (2010)
- Pitt, GR; Batt, AR; Haigh, RM; Penson, AM; Robson, PA; Rooker, DP; Tartar, AL; Trim, JE; Yea, CM; Roe, MB Non-peptide oxytocin agonists. Bioorg Med Chem Lett 14: 4585-9 (2004)
- Rosse, G Pyrazolsulfonamide agonists of oxytocin receptor. ACS Med Chem Lett 5: 1188-9 (2014)
- Brown, A; Ellis, D; Pearce, D; Ralph, M; Sciammetta, N Aryloxypyrazines as highly selective antagonists of Oxytocin. Bioorg Med Chem Lett 19: 2634-6 (2009)
- Evans, BE; Leighton, JL; Rittle, KE; Gilbert, KF; Lundell, GF; Gould, NP; Hobbs, DW; DiPardo, RM; Veber, DF; Pettibone, DJ Orally active, nonpeptide oxytocin antagonists. J Med Chem 35: 3919-27 (1992)
- Wisniewski, K; Trojnar, J; Riviere, P; Haigh, R; Yea, C; Ashworth, D; Melin, P; Nilsson, A The synthesis of a new class of oxytocin antagonists. Bioorg Med Chem Lett 9: 2801-4 (1999)
- Brown, A; Brown, L; Ellis, D; Puhalo, N; Smith, CR; Wallace, O; Watson, L Design and optimization of potent, selective antagonists of Oxytocin. Bioorg Med Chem Lett 18: 4278-81 (2008)
- Brown, A; Ellis, D; Wallace, O; Ralph, M Identification of a urea bioisostere of a triazole oxytocin antagonist. Bioorg Med Chem Lett 20: 1851-3 (2010)
- Brown, A; Ellis, D; Wallace, O; Ralph, M Identification of amide bioisosteres of triazole oxytocin antagonists. Bioorg Med Chem Lett 20: 2224-8 (2010)
- Liddle, J; Allen, MJ; Borthwick, AD; Brooks, DP; Davies, DE; Edwards, RM; Exall, AM; Hamlett, C; Irving, WR; Mason, AM; McCafferty, GP; Nerozzi, F; Peace, S; Philp, J; Pollard, D; Pullen, MA; Shabbir, SS; Sollis, SL; Westfall, TD; Woollard, PM; Wu, C; Hickey, DM The discovery of GSK221149A: a potent and selective oxytocin antagonist. Bioorg Med Chem Lett 18: 90-4 (2008)
- Evans, BE; Lundell, GF; Gilbert, KF; Bock, MG; Rittle, KE; Carroll, LA; Williams, PD; Pawluczyk, JM; Leighton, JL; Young, MB Nanomolar-affinity, non-peptide oxytocin receptor antagonists. J Med Chem 36: 3993-4005 (1994)
- Wisniewski, K; Alagarsamy, S; Galyean, R; Tariga, H; Thompson, D; Ly, B; Wisniewska, H; Qi, S; Croston, G; Laporte, R; Rivi�re, PJ; Schteingart, CD New, potent, and selective peptidic oxytocin receptor agonists. J Med Chem 57: 5306-17 (2014)
- Barton, NP; Bellenie, BR; Doran, AT; Emmons, AJ; Heer, JP; Salvagno, CM Discovery and optimisation of a potent and selective tertiary sulfonamide oxytocin antagonist. Bioorg Med Chem Lett 19: 528-32 (2008)
- Pavo, I; Slaninova, J; Klein, U; Fahrenholz, F Enhanced selectivity of oxytocin antagonists containing sarcosine in position 7. J Med Chem 37: 255-9 (1994)
- Pettibone, DJ; Clineschmidt, BV; Lis, EV; Reiss, DR; Totaro, JA; Woyden, CJ; Bock, MG; Freidinger, RM; Tung, RD; Veber, DF In vitro pharmacological profile of a novel structural class of oxytocin antagonists. J Pharmacol Exp Ther 256: 304-8 (1991)
- Muttenthaler, M; Andersson, A; de Araujo, AD; Dekan, Z; Lewis, RJ; Alewood, PF Modulating oxytocin activity and plasma stability by disulfide bond engineering. J Med Chem 53: 8585-96 (2010)
- Williams, PD; Bock, MG; Evans, BE; Freidinger, RM; Gallicchio, SN; Guidotti, MT; Jacobson, MA; Kuo, MS; Levy, MR; Lis, EV; Michelson, SR; Pawluczyk, JM; Perlow, DS; Pettibone, DJ; Quigley, AG; Reiss, DR; Salvatore, C; Stauffer, KJ; Woyden, CJ Nonpeptide oxytocin antagonists: analogs of L-371,257 with improved potency. Bioorg Med Chem Lett 9: 1311-6 (1999)
- Adachi, Y; Sakimura, K; Shimizu, Y; Nakayama, M; Terao, Y; Yano, T; Asami, T Potent and selective oxytocin receptor agonists without disulfide bridges. Bioorg Med Chem Lett 27: 2331-2335 (2017)
- Brown, A; Brown, TB; Calabrese, A; Ellis, D; Puhalo, N; Ralph, M; Watson, L Triazole oxytocin antagonists: Identification of an aryloxyazetidine replacement for a biaryl substituent. Bioorg Med Chem Lett 20: 516-20 (2010)
- Beard, R; Stucki, A; Schmitt, M; Py, G; Grundschober, C; Gee, AD; Tate, EW Building bridges for highly selective, potent and stable oxytocin and vasopressin analogs. Bioorg Med Chem 26: 3039-3045 (2018)
- Freidinger, RM; Williams, PD; Tung, RD; Bock, MG; Pettibone, DJ; Clineschmidt, BV; DiPardo, RM; Erb, JM; Garsky, VM; Gould, NP Cyclic hexapeptide oxytocin antagonists. Potency-, selectivity-, and solubility-enhancing modifications. J Med Chem 33: 1843-5 (1990)
- Katte, TA; Reekie, TA; Werry, EL; Jorgensen, WT; Boyd, R; Wong, ECN; Gulliver, DW; Connor, M; Kassiou, M Investigation of pyrazolo-sulfonamides as putative small molecule oxytocin receptor agonists. Eur J Med Chem 136: 330-333 (2017)
- Hobbs, DW; Gould, NP; Hoffman, JB; Clineschmidt, BV; Pettibone, DJ; Veber, DF; Freidinger, RM Non-peptide oxytocin antagonists: identification and synthesis of a potent camphor aminosuccinimide Bioorg Med Chem Lett 5: 119-122 (1995)
- Salituro, GM; Pettibone, DJ; Clineschmidt, BV; Williamson, JM; Zink, DL Potent, non-peptidic oxytocin receptor antagonists from a natural source Bioorg Med Chem Lett 3: 337-340 (1993)
- Wyatt, PG; Allen, MJ; Chilcott, J; Hickin, G; Miller, ND; Woollard, PM Structure-activity relationship investigations of a potent and selective benzodiazepine oxytocin antagonist. Bioorg Med Chem Lett 11: 1301-5 (2001)
- Brown, A; Brown, L; Brown, TB; Calabrese, A; Ellis, D; Puhalo, N; Smith, CR; Wallace, O; Watson, L Triazole oxytocin antagonists: identification of aryl ether replacements for a biaryl substituent. Bioorg Med Chem Lett 18: 5242-4 (2008)
- Carnazzi, E; Aumelas, A; Mouillac, B; Breton, C; Guillou, L; Barberis, C; Seyer, R Design, synthesis and pharmacological characterization of a potent radioiodinated and photoactivatable peptidic oxytocin antagonist. J Med Chem 44: 3022-30 (2001)
- Williams, PD; Bock, MG; Tung, RD; Garsky, VM; Perlow, DS; Erb, JM; Lundell, GF; Gould, NP; Whitter, WL; Hoffman, JB Development of a novel class of cyclic hexapeptide oxytocin antagonists based on a natural product. J Med Chem 35: 3905-18 (1992)
- Wyatt, PG; Allen, MJ; Chilcott, J; Foster, A; Livermore, DG; Mordaunt, JE; Scicinski, J; Woollard, PM Identification of potent and selective oxytocin antagonists. Part 1: indole and benzofuran derivatives. Bioorg Med Chem Lett 12: 1399-404 (2002)
- Smith, AL; Freeman, SM; Voll, RJ; Young, LJ; Goodman, MM Investigation of an F-18 oxytocin receptor selective ligand via PET imaging. Bioorg Med Chem Lett 23: 5415-20 (2013)
- Structure-Based Design of Glycosylated Oxytocin Analogues with Improved Selectivity and Antinociceptive Activity.
- Tóth, GK; Bakos, K; Penke, B; Pávó, I; Varga, C; Török, G; Péter, A; Fülöp, F Synthesis of oxytocin antagonists containing conformationally constrained amino acids in position 2. Bioorg Med Chem Lett 9: 667-72 (1999)
- Sciabola, S; Goetz, GH; Bai, G; Rogers, BN; Gray, DL; Duplantier, A; Fonseca, KR; Vanase-Frawley, MA; Kablaoui, NM Systematic N-methylation of oxytocin: Impact on pharmacology and intramolecular hydrogen bonding network. Bioorg Med Chem 24: 3513-20 (2016)
- Quattropani, A; Dorbais, J; Covini, D; Pittet, PA; Colovray, V; Thomas, RJ; Coxhead, R; Halazy, S; Scheer, A; Missotten, M; Ayala, G; Bradshaw, C; De Raemy-Schenk, AM; Nichols, A; Cirillo, R; Tos, EG; Giachetti, C; Golzio, L; Marinelli, P; Church, DJ; Barberis, C; Chollet, A; Schwarz, MK Discovery and development of a new class of potent, selective, orally active oxytocin receptor antagonists. J Med Chem 48: 7882-905 (2005)
- Manning, M; Cheng, LL; Klis, WA; Balaspiri, L; Olma, A; Sawyer, WH; Wo, NC; Chan, WY Effects of a D-Cys6/L-Cys6 interchange in nonselective and selective vasopressin and oxytocin antagonists. J Med Chem 38: 1762-9 (1995)
- Jorgensen, WT; Gulliver, DW; Werry, EL; Reekie, T; Connor, M; Kassiou, M Flexible analogues of WAY-267,464: Synthesis and pharmacology at the human oxytocin and vasopressin 1a receptors. Eur J Med Chem 108: 730-40 (2016)
- Wyatt, PG; Allen, MJ; Chilcott, J; Gardner, CJ; Livermore, DG; Mordaunt, JE; Nerozzi, F; Patel, M; Perren, MJ; Weingarten, GG; Shabbir, S; Woollard, PM; Zhou, P Identification of potent and selective oxytocin antagonists. Part 2: further investigation of benzofuran derivatives. Bioorg Med Chem Lett 12: 1405-11 (2002)
- Frantz, MC; Rodrigo, J; Boudier, L; Durroux, T; Mouillac, B; Hibert, M Subtlety of the structure-affinity and structure-efficacy relationships around a nonpeptide oxytocin receptor agonist. J Med Chem 53: 1546-62 (2010)
- Fragiadaki, M; Magafa, V; Borovicková, L; Slaninová, J; Cordopatis, P Synthesis and biological activity of oxytocin analogues containing conformationally-restricted residues in position 7. Eur J Med Chem 42: 799-806 (2007)
- Terrillon, S; Cheng, LL; Stoev, S; Mouillac, B; Barberis, C; Manning, M; Durroux, T Synthesis and characterization of fluorescent antagonists and agonists for human oxytocin and vasopressin V(1)(a) receptors. J Med Chem 45: 2579-88 (2002)
- Borthwick, AD; Davies, DE; Exall, AM; Livermore, DG; Sollis, SL; Nerozzi, F; Allen, MJ; Perren, M; Shabbir, SS; Woollard, PM; Wyatt, PG 2,5-diketopiperazines as potent, selective, and orally bioavailable oxytocin antagonists. 2. Synthesis, chirality, and pharmacokinetics. J Med Chem 48: 6956-69 (2005)
- Smith, AL; Walum, H; Connor-Stroud, F; Freeman, SM; Inoue, K; Parr, LA; Goodman, MM; Young, LJ An evaluation of central penetration from a peripherally administered oxytocin receptor selective antagonist in nonhuman primates. Bioorg Med Chem 25: 305-315 (2017)
- Jorgensen, WT; Gulliver, DW; Katte, TA; Werry, EL; Reekie, TA; Connor, M; Kassiou, M Conformationally rigid derivatives of WAY-267,464: Synthesis and pharmacology at the human oxytocin and vasopressin-1a receptors. Eur J Med Chem 143: 1644-1656 (2018)
- Corbani, M; Trueba, M; Stoev, S; Murat, B; Mion, J; Boulay, V; Guillon, G; Manning, M Design, synthesis, and pharmacological characterization of fluorescent peptides for imaging human V1b vasopressin or oxytocin receptors. J Med Chem 54: 2864-77 (2011)
- Borthwick, AD; Davies, DE; Exall, AM; Hatley, RJ; Hughes, JA; Irving, WR; Livermore, DG; Sollis, SL; Nerozzi, F; Valko, KL; Allen, MJ; Perren, M; Shabbir, SS; Woollard, PM; Price, MA 2,5-diketopiperazines as potent, selective, and orally bioavailable oxytocin antagonists. 3. Synthesis, pharmacokinetics, and in vivo potency. J Med Chem 49: 4159-70 (2006)
- Busnelli, M; Kleinau, G; Muttenthaler, M; Stoev, S; Manning, M; Bibic, L; Howell, LA; McCormick, PJ; Di Lascio, S; Braida, D; Sala, M; Rovati, GE; Bellini, T; Chini, B Design and Characterization of Superpotent Bivalent Ligands Targeting Oxytocin Receptor Dimers via a Channel-Like Structure. J Med Chem 59: 7152-66 (2016)
- Ichinose, W; Cherepanov, SM; Shabalova, AA; Yokoyama, S; Yuhi, T; Yamaguchi, H; Watanabe, A; Yamamoto, Y; Okamoto, H; Horike, S; Terakawa, J; Daikoku, T; Watanabe, M; Mano, N; Higashida, H; Shuto, S Development of a Highly Potent Analogue and a Long-Acting Analogue of Oxytocin for the Treatment of Social Impairment-Like Behaviors. J Med Chem 62: 3297-3310 (2019)
- Bellenie, BR; Barton, NP; Emmons, AJ; Heer, JP; Salvagno, C Discovery and optimization of highly ligand-efficient oxytocin receptor antagonists using structure-based drug design. Bioorg Med Chem Lett 19: 990-4 (2009)
- PubChem, PC Fluorescence-based cell-based high throughput dose response assay for agonists of the Oxytocin Receptor (OXTR) PubChem Bioassay (2010)
- PubChem, PC Fluorescence-based cell-based high throughput dose response assay for potentiators of Oxytocin Receptor (OXTR) PubChem Bioassay (2010)
- Frantz, MC; Pellissier, LP; Pflimlin, E; Loison, S; Gand�a, J; Marsol, C; Durroux, T; Mouillac, B; Becker, JAJ; Le Merrer, J; Valencia, C; Villa, P; Bonnet, D; Hibert, M LIT-001, the First Nonpeptide Oxytocin Receptor Agonist that Improves Social Interaction in a Mouse Model of Autism. J Med Chem 61: 8670-8692 (2018)
- Borthwick, AD; Liddle, J; Davies, DE; Exall, AM; Hamlett, C; Hickey, DM; Mason, AM; Smith, IE; Nerozzi, F; Peace, S; Pollard, D; Sollis, SL; Allen, MJ; Woollard, PM; Pullen, MA; Westfall, TD; Stanislaus, DJ Pyridyl-2,5-diketopiperazines as potent, selective, and orally bioavailable oxytocin antagonists: synthesis, pharmacokinetics, and in vivo potency. J Med Chem 55: 783-96 (2012)
- Chen, L; Zoulíková, I; Slaninová, J; Barany, G Synthesis and pharmacology of novel analogues of oxytocin and deaminooxytocin: directed methods for the construction of disulfide and trisulfide bridges in peptides. J Med Chem 40: 864-76 (1997)
- Smith, AL; Freeman, SM; Voll, RJ; Young, LJ; Goodman, MM Carbon-11 N-methyl alkylation of L-368,899 and in vivo PET imaging investigations for neural oxytocin receptors. Bioorg Med Chem Lett 23: 902-6 (2013)
- Pflimlin, E; Zhou, Z; Amso, Z; Fu, Q; Lee, C; Muppiddi, A; Joseph, SB; Nguyen-Tran, V; Shen, W Engineering a Potent, Long-Acting, and Periphery-Restricted Oxytocin Receptor Agonist with Anorexigenic and Body Weight Reducing Effects. J Med Chem 63: 382-390 (2020)
- Evans, B; Pipe, A; Clark, L; Banks, M Identification of a potent and selective oxytocin antagonist, from screening a fully encoded differential release combinatorial chemical library. Bioorg Med Chem Lett 11: 1297-300 (2001)
- Karpenko, IA; Margathe, JF; Rodriguez, T; Pflimlin, E; Dupuis, E; Hibert, M; Durroux, T; Bonnet, D Selective nonpeptidic fluorescent ligands for oxytocin receptor: design, synthesis, and application to time-resolved FRET binding assay. J Med Chem 58: 2547-52 (2015)
- Smith, AL; Freeman, SM; Stehouwer, JS; Inoue, K; Voll, RJ; Young, LJ; Goodman, MM Synthesis and evaluation of C-11, F-18 and I-125 small molecule radioligands for detecting oxytocin receptors. Bioorg Med Chem 20: 2721-38 (2012)
- Albizu, L; Teppaz, G; Seyer, R; Bazin, H; Ansanay, H; Manning, M; Mouillac, B; Durroux, T Toward efficient drug screening by homogeneous assays based on the development of new fluorescent vasopressin and oxytocin receptor ligands. J Med Chem 50: 4976-85 (2007)
- Kuo, MS; Bock, MG; Freidinger, RM; Guidotti, MT; Lis, EV; Pawluczyk, JM; Perlow, DS; Pettibone, DJ; Quigley, AG; Reiss, DR; Williams, PD; Woyden, CJ Nonpeptide oxytocin antagonists: potent, orally bioavailable analogs of L-371,257 containing a 1-R-(pyridyl)ethyl ether terminus. Bioorg Med Chem Lett 8: 3081-6 (1999)
- Bock, MG; DiPardo, RM; Williams, PD; Pettibone, DJ; Clineschmidt, BV; Ball, RG; Veber, DF; Freidinger, RM Receptor ligands which bind the oxytocin receptor with selectivity and high affinity. Chemical modification of a Streptomyces silvensis derived cyclic hexapeptide. J Med Chem 33: 2321-3 (1990)
- Cherepanov, SM; Miura, R; Shabalova, AA; Ichinose, W; Yokoyama, S; Fukuda, H; Watanabe, M; Higashida, H; Shuto, S Synthesis of oxytocin derivatives lipidated via a carbonate or carbamate linkage as a long-acting therapeutic agent for social impairment-like behaviors. Bioorg Med Chem 27: 3358-3363 (2019)
- PubChem, PC Counterscreen for vasopressin 1 receptor (V1R) agonists: Fluorescence-based cell-based high throughput dose response assay for agonists of the Oxytocin Receptor (OXTR) PubChem Bioassay (2010)
- PubChem, PC Counterscreen for Oxytocin Receptor (OXTR) agonists: Fluorescence-based cell-based high throughput dose response assay to identify agonists of the vasopressin 1 receptor (V1R) PubChem Bioassay (2010)
- Bélec, L; Slaninova, J; Lubell, WD A study of the relationship between biological activity and prolyl amide isomer geometry in oxytocin using 5-tert-butylproline to augment the Cys(6)-Pro(7) amide cis-isomer population. J Med Chem 43: 1448-55 (2000)
- Williams, PD; Clineschmidt, BV; Erb, JM; Freidinger, RM; Guidotti, MT; Lis, EV; Pawluczyk, JM; Pettibone, DJ; Reiss, DR; Veber, DF 1-(1-[4-[(N-acetyl-4-piperidinyl)oxy]-2-methoxybenzoyl]piperidin-4- yl)-4H-3,1-benzoxazin-2(1H)-one (L-371,257): a new, orally bioavailable, non-peptide oxytocin antagonist. J Med Chem 38: 4634-6 (1995)
- Cirillo, R; Gillio Tos, E; Schwarz, MK; Quattropani, A; Scheer, A; Missotten, M; Dorbais, J; Nichols, A; Borrelli, F; Giachetti, C; Golzio, L; Marinelli, P; Thomas, RJ; Chevillard, C; Laurent, F; Portet, K; Barberis, C; Chollet, A Pharmacology of (2S,4Z)-N-[(2S)-2-hydroxy-2-phenylethyl]-4-(methoxyimino) -1-[(2'-methyl[1,1'-biphenyl]-4-yl)carbonyl]-2-pyrrolidinecarboxamide, a new potent and selective nonpeptide antagonist of the oxytocin receptor. J Pharmacol Exp Ther 306: 253-61 (2003)
- Bell, IM; Erb, JM; Freidinger, RM; Gallicchio, SN; Guare, JP; Guidotti, MT; Halpin, RA; Hobbs, DW; Homnick, CF; Kuo, MS; Lis, EV; Mathre, DJ; Michelson, SR; Pawluczyk, JM; Pettibone, DJ; Reiss, DR; Vickers, S; Williams, PD; Woyden, CJ Development of orally active oxytocin antagonists: studies on 1-(1-[4-[1-(2-methyl-1-oxidopyridin-3-ylmethyl)piperidin-4-yloxy]-2- methoxybenzoyl]piperidin-4-yl)-1,4-dihydrobenz[d][1,3]oxazin-2-one (L-372,662) and related pyridines. J Med Chem 41: 2146-63 (1998)
- Williams, PD; Anderson, PS; Ball, RG; Bock, MG; Carroll, L; Chiu, SH; Clineschmidt, BV; Culberson, JC; Erb, JM; Evans, BE 1-((7,7-Dimethyl-2(S)-(2(S)-amino-4-(methylsulfonyl)butyramido)bicyclo [2.2.1]-heptan-1(S)-yl)methyl)sulfonyl)-4-(2-methylphenyl)piperaz ine (L-368,899): an orally bioavailable, non-peptide oxytocin antagonist with potential utility for managing preterm labor. J Med Chem 37: 565-71 (1994)
- ChEMBL_149053 (CHEMBL857864) Inhibition of binding of [3H]oxytocin with human oxytocin receptor
- ChEMBL_321073 (CHEMBL885138) Displacement of [3H]oxytocin from human Oxytocin receptor
- ChEMBL_652651 (CHEMBL1225854) Displacement of [3H]oxytocin from human oxytocin receptor
- ChEMBL_661588 (CHEMBL1251522) Displacement of [3H]-oxytocin from human oxytocin receptor
- ChEMBL_727047 (CHEMBL1686971) Displacement of [3H]oxytocin from human oxytocin receptor
- ChEMBL_821157 (CHEMBL2039699) Displacement of [3H]oxytocin from human oxytocin receptor
- ChEMBL_149054 (CHEMBL761408) Displacement of 3[H]oxytocin from human oxytocin receptor
- ChEMBL_149058 (CHEMBL761411) Inhibition of binding of [3H]oxytocin to rat uterine oxytocin receptor
- ChEMBL_149182 (CHEMBL762745) Inhibition of [3H]- oxytocin binding to rat uterine Oxytocin receptor
- ChEMBL_661536 (CHEMBL1253100) Displacement of [3H]-oxytocin from oxytocin receptor in human uterus tissue
- ChEMBL_661537 (CHEMBL1253101) Displacement of [3H]-oxytocin from oxytocin receptor in rat uterus tissue
- ChEMBL_449794 (CHEMBL898900) Displacement of [3H]oxytocin from human oxytocin receptor expressed in CHO cells
- ChEMBL_661552 (CHEMBL1253116) Displacement of [3H]-oxytocin from human oxytocin receptor expressed in CHO cells
- ChEMBL_661556 (CHEMBL1253120) Displacement of [3H]-oxytocin from rat oxytocin receptor expressed in CHO cells
- ChEMBL_748692 (CHEMBL1780545) Displacement of [3H]oxytocin from human oxytocin receptor expressed in CHO cells
- ChEMBL_145030 (CHEMBL753724) Inhibition of radioligand [3H]oxytocin binding at the oxytocin (OT) receptor in rat uterine tissue
- ChEMBL_149056 (CHEMBL761410) Compound was tested for its ability to displace [3H]oxytocin from human Oxytocin receptor
- ChEMBL_661577 (CHEMBL1253141) Displacement of [3H]-oxytocin from human oxytocin receptor expressed in HEK293-EBNA cells
- ChEMBL_661578 (CHEMBL1253142) Displacement of [3H]-oxytocin from rat oxytocin receptor expressed in HEK293-EBNA cells
- ChEMBL_821172 (CHEMBL2039714) Antagonist activity at human oxytocin receptor assessed as inhibition of oxytocin binding by FLIPR analysis
- ChEBML_149062 Compound was tested for its ability to inhibit binding of Oxytocin to its Oxytocin receptor in rat uterine tissue
- ChEMBL_748741 (CHEMBL1780594) Displacement of radiolabeled oxytocin from human oxytocin receptor expressed in CHO cells by scintillation counting
- ChEMBL_149060 (CHEMBL760424) Compound was evaluated for half-maximal inhibition of binding of [3H]oxytocin to Oxytocin receptor in rat uterine tissue
- ChEMBL_149174 (CHEMBL762089) Binding affinity against Oxytocin receptor was determined in rat uterine membrane using radioligand [3H]oxytocin
- ChEMBL_515100 (CHEMBL1035608) Displacement of [3H]oxytocin from human oxytocin receptor expressed in CHO cells by filtration binding assay
- ChEMBL_813470 (CHEMBL2019478) Displacement of [3H]oxytocin from human oxytocin receptor after 1.5 hrs by liquid scintillation counting
- ChEBML_149046 Binding affinity for human oxytocin receptor
- ChEMBL_515097 (CHEMBL1035605) Binding affinity to oxytocin receptor
- ChEMBL_561007 (CHEMBL1015247) Binding affinity to oxytocin receptor
- ChEMBL_651959 (CHEMBL1227632) Antagonist activity at oxytocin receptor
- ChEMBL_864207 (CHEMBL2175259) Binding affinity to oxytocin receptor
- ChEMBL_540354 (CHEMBL1025697) Displacement of 3H-oxytocin from human recombinant oxytocin receptor expressed in CHO cells by filtration binding assay
- ChEMBL_590266 (CHEMBL1052801) Displacement of [3H]oxytocin from human oxytocin receptor expressed in CHO-K1 cells by Packard Topcount scintillation counter
- ChEMBL_149044 (CHEMBL762237) Binding affinity for human oxytocin receptor
- ChEMBL_149203 (CHEMBL759542) Antagonistic activity against Oxytocin receptor
- ChEMBL_149204 (CHEMBL759543) Binding affinity towards oxytocin receptor
- ChEMBL_220998 (CHEMBL821744) Binding affinity against oxytocin receptor
- ChEMBL_304139 (CHEMBL840256) Effective concentration for human Oxytocin receptor
- ChEMBL_465428 (CHEMBL946012) Binding affinity to human oxytocin receptor
- ChEMBL_465438 (CHEMBL946022) Binding affinity to recombinant oxytocin receptor
- ChEMBL_326543 (CHEMBL863363) Inhibitory activity against human Oxytocin induced intracellular Calcium mobilization in human Oxytocin receptor transfected HEK293-EBNA cells
- ChEMBL_1473360 (CHEMBL3419320) Displacement of [3H]-oxytocin from human oxytocin receptor expressed in HEK cell membranes after 1 hr by scintillation proximity assay
- ChEBML_149179 Binding affinity for rat uterine oxytocin receptor (rOTr)
- ChEBML_149183 Binding affinity against oxytocin receptor in rat uterus
- ChEMBL_1555978 (CHEMBL3767486) Displacement of [3H]-oxytocin from human oxytocin receptor expressed in HEK293 cells after 90 mins by microbeta 2 microplate reader analysis
- ChEBML_149042 Inhibitory concentration required for antagonist activity against oxytocin receptor
- ChEMBL_149175 (CHEMBL762090) Binding affinity for rat oxytocin receptor (OT-R)
- ChEMBL_1474017 (CHEMBL3419777) Competitive binding to human oxytocin receptor by radioligand binding assay
- ChEMBL_149043 (CHEMBL762236) Binding affinity for cloned human oxytocin receptor (OT-R)
- ChEMBL_321075 (CHEMBL885140) Displacement of [3H]oxytocin from human V2 vasopressin receptor
- ChEMBL_321076 (CHEMBL872156) Displacement of [3H]oxytocin from human V1b vasopressin receptor
- ChEMBL_321077 (CHEMBL872157) Displacement of [3H]oxytocin from human V1a vasopressin receptor
- ChEMBL_1555992 (CHEMBL3767656) Displacement of [Tyrosyl-2,6-3H]-Oxytocin from recombinant human oxytocin receptor expressed in CHO-DUKX-A2 cells after 180 mins by liquid scintillation counting analysis
- ChEBML_193005 Binding affinity against oxytocin receptor (rOTr) in DES pretreated rat uterine
- ChEMBL_1474016 (CHEMBL3419776) Competitive binding to oxytocin receptor (unknown origin) by radioligand binding assay
- ChEMBL_149055 (CHEMBL761409) Ability to displace [3H]oxytocin from human OT receptor (hOT)
- ChEMBL_149171 (CHEMBL760342) Displacement of [3H]OT from binding to oxytocin receptor of rat uterus
- ChEMBL_697994 (CHEMBL1633054) Displacement of [3H]OT from oxytocin receptor expressed in COS1 cells
- ChEMBL_1730147 (CHEMBL4145425) Antagonist activity at human OXTR expressed in CHOK1 cells assessed as oxytocin-induced beta-arrestin recruitment pre-incubated for 30 mins before oxytocin stimulation for 90 or 180 mins by chemiluminescence method
- ChEBML_149040 Compound was evaluated for its dissociation constant (Kd) to guinea pig myometrial Oxytocin receptor
- ChEBML_149059 Half-maximal inhibition of binding of [3H]oxytocin to OT receptor in rat uterine tissue
- ChEBML_149178 Binding affinity towards rat uterine receptor was determined using [3H]oxytocin as radioligand
- ChEBML_149202 Compound was tested for displacement of 3[H] oxytocin from rat OT receptor (in vitro)
- ChEMBL_149063 (CHEMBL760427) Concentration required to displace 50% of [3H]oxytocin from rat uterine receptor.
- ChEMBL_1766336 (CHEMBL4201583) Displacement of [3H]-oxytocin from OTR receptor in human USMC cell membranes
- ChEMBL_446244 (CHEMBL895350) Displacement of [3H]AVP from human oxytocin receptor expressed in CHO cells
- ChEMBL_515099 (CHEMBL1035607) Antagonist activity at human oxytocin receptor expressed in CHO cells by FLIPR assay
- ChEMBL_609611 (CHEMBL1065226) Displacement of [3H]AVP from human oxytocin receptor expressed in CHO cells
- ChEBML_1555980 Antagonist activity at human oxytocin receptor expressed in HEK293 cells assessed as inhibition of oxytocin induced IP1 accumulation pretreated for 30 mins measured 1 hr post Ab-Cryptate and IP1-d2 addition by HTRF assay
- Oxytocin Receptor Antagonist Assay This assay was performed in CHEM-1 cells expressing the human Oxytocin Receptor (hOTR) to determine the inhibitory effect of the compounds of the invention on the human Oxytocin receptor. Oxytocin evokes an increase in intracellular calcium in CHEM-1-hOTR cells which is measured in a fluorescence assay on the FLIPRTETRA using calcium sensitive dyes. Test compounds were assessed for their ability to affect the magnitude of the response to oxytocin, with antagonists showing a concentration-dependent reduction in the oxytocin-mediated fluorescence. Compounds displaying potency at the vasopressin V1a receptor of <100 nM were progressed to selectivity testing against hOTR and were tested in triplicate in a 10-point, 1:3 dilution series starting at a nominal concentration of 3 μM in the assay.CHEM-1-hOTR ready was used to assay frozen cells (Eurofins #HTS090RTA) which are supplied with a proprietary Media Component.
- ChEBML_149050 Binding affinity against cloned human oxytocin receptor from human embryonic kidney cells
- ChEMBL_149040 (CHEMBL762233) Compound was evaluated for its dissociation constant (Kd) to guinea pig myometrial Oxytocin receptor
- ChEMBL_149178 (CHEMBL882439) Binding affinity towards rat uterine receptor was determined using [3H]oxytocin as radioligand
- ChEMBL_149202 (CHEMBL759541) Compound was tested for displacement of 3[H] oxytocin from rat OT receptor (in vitro)
- ChEMBL_540347 (CHEMBL1025690) Antagonist activity at human recombinant oxytocin receptor expressed in CHO cells by FLIPR assay
- ChEMBL_578145 (CHEMBL1059543) Antagonist activity at human cloned oxytocin receptor by cell based beta-lactamase assay
- ChEMBL_604218 (CHEMBL1050363) Antagonist activity at human cloned oxytocin receptor by cell based beta lactamase assay
- ChEMBL_615224 (CHEMBL1115874) Antagonist activity at human oxytocin receptor expressed in CHO cells by beta lactamase assay
- ChEMBL_813468 (CHEMBL2019476) Displacement of [125I]-OVTA from prairie vole Oxytocin receptor after 72 hrs
- ChEMBL_1555980 (CHEMBL3767488) Antagonist activity at human oxytocin receptor expressed in HEK293 cells assessed as inhibition of oxytocin induced IP1 accumulation pretreated for 30 mins measured 1 hr post Ab-Cryptate and IP1-d2 addition by HTRF assay
- ChEMBL_148874 (CHEMBL882495) Inhibition of radioligand [3H]OT binding to oxytocin receptor (OT) in guinea pig myometrium membrane
- ChEMBL_326537 (CHEMBL863355) Displacement of [125I]OVTA antagonist from human oxytocin receptor expressed in HEK293-EBNA cells
- ChEMBL_326541 (CHEMBL863361) Displacement of [125I]OVTA antagonist from rat oxytocin receptor expressed in HEK293-EBNA cells
- ChEMBL_513959 (CHEMBL974600) Antagonist activity at human cloned oxytocin receptor by cell based beta lactamase reporter assay
- ChEMBL_535596 (CHEMBL987070) Antagonist activity at human oxytocin receptor by cell based beta-lactamase reporter gene assay
- ChEMBL_540346 (CHEMBL1025689) Binding affinity to human recombinant oxytocin receptor expressed in CHO cells by fluorescence polarization assay
- ChEMBL_609617 (CHEMBL1065232) Activity at human oxytocin receptor expressed in CHO cells by NFAT-luciferase gene reporter assay
- ChEMBL_697993 (CHEMBL1633053) Activity at oxytocin receptor expressed in COS1 cells assessed as IP-one generation by HTRF assay
- ChEBML_149051 In vivo antagonistic activity against cloned human oxytocin receptor over-expressed in a stable HEK293 cell line
- ChEMBL_1431801 (CHEMBL3385097) Agonist activity at human oxytocin receptor expressed CHO cells assessed as calcium flux by FLIPR method
- ChEMBL_743722 (CHEMBL1767536) Displacement of [3H]AVP from human oxytocin receptor expressed CHO cells after 60 mins
- ChEMBL_864195 (CHEMBL2175247) Displacement of [3H]-AVP from human oxytocin receptor expressed in CHO cells after 30 mins
- ChEMBL_1711200 (CHEMBL4121249) Antagonist activity at human mammary gland oxytocin receptor expressed in CHO cells assessed as inhibition of agonist-induced intracellular calcium flux preincubated for 20 mins followed by oxytocin addition measured for 5 mins by Fluo-4-AM dye based FLIPR assay
- ChEMBL_1474007 (CHEMBL3419767) Binding affinity to SNAP-tagged oxytocin receptor (unknown origin) expressed in HEK293 cells by TR-FRET assay
- ChEMBL_149051 (CHEMBL761406) In vivo antagonistic activity against cloned human oxytocin receptor over-expressed in a stable HEK293 cell line
- ChEMBL_515103 (CHEMBL1024625) Displacement of [3H]oxytocin from human vasopressin V2 receptor expressed in CHO cells by filtration binding assay
- ChEMBL_373748 (CHEMBL870969) Agonist activity at human OTR expressed in CHO cells assessed as inhibition of oxytocin-induced calcium mobilization by FLIPR assay
- ChEMBL_1726373 (CHEMBL4141651) Displacement of [3H]-oxytocin from human OTR expressed in HEK293 cell membranes after 90 mins by radioligand binding assay
- ChEMBL_1766328 (CHEMBL4201575) Displacement of [tyrosyl-2,6-3H] oxytocin from OTR (unknown origin) expressed in HEK293 cell membranes incubated for 60 mins
- ChEMBL_1451726 (CHEMBL3366919) Agonist activity at human oxytocin receptor expressed in CHO-K1 cells after 5 hrs by firefly luciferase reporter gene assay
- ChEMBL_149047 (CHEMBL761402) Binding affinity was evaluated by measuring the displacement of [3H]OT (oxytocin) from specific binding sites in uterine tissue obtained from human
- ChEMBL_149048 (CHEMBL761403) Binding affinity was evaluated by measuring the displacement of [3H]-OT (oxytocin) from specific binding sites in uterine tissue obtained from human.
- ChEMBL_149052 (CHEMBL761407) The inhibition constant (Ki(nM)) by displacement of [125I]- HO-LVA radiolabeled ligand using membranes of CHO cells of human Oxytocin receptor
- ChEMBL_149180 (CHEMBL762743) Binding affinity was evaluated by measuring the displacement of [3H]-OT (oxytocin) from specific binding sites in uterine tissue obtained from rats
- ChEMBL_1651553 (CHEMBL4000808) Displacement of [3H]Oxytocin from human OXTR expressed in CHO cell membranes after 90 mins by liquid scintillation counting method
- ChEMBL_1724090 (CHEMBL4139368) Displacement of [3H]-oxytocin from human OTR expressed in HEK293 cell membranes after 90 mins by microbeta scintillation counting method
- ChEMBL_149045 (CHEMBL762238) Binding affinity against human oxytocin receptor was determined by using plasma membranes from CHO cells stably transfected with VP/OT receptors
- ChEMBL_149057 (CHEMBL873306) Binding affinity against human oxytocin receptor was determined by using plasma membranes from CHO cells stably transfected with VP/OT receptors
- ChEMBL_1555993 (CHEMBL3767657) Agonist activity at recombinant human oxytocin receptor expressed in CHO-DUKX-A2 cells assessed as change in intracellular calcium level by FLIPR assay
- ChEBML_1681637 Displacement of [Tyrosyl-2,6-3H]Oxytocin from recombinant mouse OTR expressed in CHO-K1 cells after 180 mins by liquid scintillation counting method
- ChEMBL_1711191 (CHEMBL4121240) Displacement of [3H]OT from human mammary gland oxytocin receptor expressed in HEK293 cell membrane after 1 hr by scintillation counting method
- ChEMBL_1474013 (CHEMBL3419773) Agonist activity at SNAP-tagged oxytocin receptor (unknown origin) expressed in HEK293 cells assessed as intracellular calcium flux after 3 mins by fluorescence assay
- ChEMBL_1583335 (CHEMBL3816398) Displacement of [3H]8-arginine-vasopressin from human oxytocin receptor expressed in CHO cell membrane incubated for 1 hr by liquid scintillation counting method
- ChEMBL_700576 (CHEMBL1648395) Antagonist activity at recombinant oxytocin receptor expressed in CHO cells assessed as inhibition of vasopressin-induced calcium release after 10 mins by Fluo4-AM staining
- ChEMBL_743728 (CHEMBL1767542) Agonist activity at human oxytocin receptor expressed CHO cells assessed as induction of phospholipase C activity after 15 mins by inositol phosphate accumulation assay
- ChEBML_1583333 Agonist activity at human oxytocin receptor expressed in CHO cells assessed as increase in intracellular calcium flux measured for 90 sec by fluo-4 dye based FLIPR assay
- ChEMBL_1474018 (CHEMBL3419778) Binding affinity to SNAP-tagged oxytocin receptor (unknown origin) expressed in HEK293 cells assessed as association constant after 5 to 120 mins by TR-FRET assay
- ChEMBL_1474023 (CHEMBL3419994) Binding affinity to SNAP-tagged oxytocin receptor (unknown origin) expressed in HEK293 cells assessed as dissociation constant after 5 to 120 mins by TR-FRET assay
- ChEMBL_1681633 (CHEMBL4031910) Displacement of [Tyrosyl-2,6-3H]Oxytocin from recombinant human OTR expressed in CHO-DUKX-A2 cells after 180 mins by liquid scintillation counting method
- ChEMBL_1583333 (CHEMBL3816396) Agonist activity at human oxytocin receptor expressed in CHO cells assessed as increase in intracellular calcium flux measured for 90 sec by fluo-4 dye based FLIPR assay
- ChEMBL_1555979 (CHEMBL3767487) Agonist activity at human oxytocin receptor expressed in HEK293 cells assessed as increase in IP1 accumulation preincubated for 1 hr followed by addition of Ab-Cryptate and IP1-d2 by HTRF assay
- ChEMBL_1711194 (CHEMBL4121243) Agonist activity at human mammary gland oxytocin receptor expressed in CHO cells assessed as increase in intracellular calcium flux measured for 5 mins by Fluo-4-AM dye based FLIPR assay
- ChEMBL_1766322 (CHEMBL4201569) Displacement of [Tyrosyl-2,6-3H]oxytocin from recombinant human OTR expressed in CHO-DUKX-A2 cells incubated for 180 mins by scintillation counting based whole cell radioligand binding assay
- ChEMBL_1933582 (CHEMBL4479234) Agonist activity at human oxytocin receptor expressed in HEK293 cells coexpressing Rluc8-tagged Galphaq, N-terminal GFP-tagged Ggamma2 and Gbeta1 protein incubated for 2 mins by Gq protein activation based BRET assay
- ChEMBL_1933586 (CHEMBL4479238) Agonist activity at human oxytocin receptor C47A mutant expressed in HEK293 cells coexpressing Rluc8-tagged Galphaq, N-terminal GFP-tagged Ggamma2 and Gbeta1 protein incubated for 2 mins by Gq protein activation based BRET assay
- ChEMBL_1933583 (CHEMBL4479235) Agonist activity at human oxytocin receptor high affinity site expressed in HEK293 cells coexpressing Rluc8-tagged Galphaq, N-terminal GFP-tagged Ggamma2 and Gbeta1 protein incubated for 2 mins by Gq protein activation based BRET assay
- ChEMBL_1933584 (CHEMBL4479236) Agonist activity at human oxytocin receptor low affinity site expressed in HEK293 cells coexpressing Rluc8-tagged Galphaq, N-terminal GFP-tagged Ggamma2 and Gbeta1 protein incubated for 2 mins by Gq protein activation based BRET assay
- ChEMBL_1933591 (CHEMBL4479243) Agonist activity at human oxytocin receptor V43A/C47A double mutant expressed in HEK293 cells coexpressing Rluc8-tagged Galphaq, N-terminal GFP-tagged Ggamma2 and Gbeta1 protein incubated for 2 mins by Gq protein activation based BRET assay
- ChEMBL_1933594 (CHEMBL4479246) Agonist activity at human oxytocin receptor expressed in HEK293 cells coexpressing Rluc8-tagged Galphaq, N-terminal GFP-tagged Ggamma2 and Gbeta1 protein incubated for 2 mins in presence of TMH1-TAT peptide by Gq protein activation based BRET assay
- ChEMBL_1933595 (CHEMBL4479247) Agonist activity at human oxytocin receptor expressed in HEK293 cells coexpressing Rluc8-tagged Galphaq, N-terminal GFP-tagged Ggamma2 and Gbeta1 protein incubated for 2 mins in presence of TMH5-TAT peptide by Gq protein activation based BRET assay
- ChEMBL_1933589 (CHEMBL4479241) Agonist activity at human oxytocin receptor C47A mutant high affinity site expressed in HEK293 cells coexpressing Rluc8-tagged Galphaq, N-terminal GFP-tagged Ggamma2 and Gbeta1 protein incubated for 2 mins by Gq protein activation based BRET assay
- ChEMBL_1933590 (CHEMBL4479242) Agonist activity at human oxytocin receptor C47A mutant low affinity site expressed in HEK293 cells coexpressing Rluc8-tagged Galphaq, N-terminal GFP-tagged Ggamma2 and Gbeta1 protein incubated for 2 mins by Gq protein activation based BRET assay
- ChEMBL_1933592 (CHEMBL4479244) Agonist activity at human oxytocin receptor V43A/C47A double mutant high affinity site expressed in HEK293 cells coexpressing Rluc8-tagged Galphaq, N-terminal GFP-tagged Ggamma2 and Gbeta1 protein incubated for 2 mins by Gq protein activation based BRET assay
- ChEMBL_1933593 (CHEMBL4479245) Agonist activity at human oxytocin receptor V43A/C47A double mutant low affinity site expressed in HEK293 cells coexpressing Rluc8-tagged Galphaq, N-terminal GFP-tagged Ggamma2 and Gbeta1 protein incubated for 2 mins by Gq protein activation based BRET assay
- ChEMBL_1933602 (CHEMBL4479254) Agonist activity at human oxytocin receptor high affinity site expressed in HEK293 cells coexpressing Rluc8-tagged Galphaq, N-terminal GFP-tagged Ggamma2 and Gbeta1 protein incubated for 2 mins in presence of TMH5-TAT peptide by Gq protein activation based BRET assay
- ChEMBL_1933603 (CHEMBL4479255) Agonist activity at human oxytocin receptor low affinity site expressed in HEK293 cells coexpressing Rluc8-tagged Galphaq, N-terminal GFP-tagged Ggamma2 and Gbeta1 protein incubated for 2 mins in presence of TMH5-TAT peptide by Gq protein activation based BRET assay
- Fluorescence-based cell-based high throughput dose response assay for agonists of the Oxytocin Receptor (OXTR) Source (MLPCN Center Name): The Scripps Research Institute Molecular Screening Center (SRIMSC) Center Affiliation: The Scripps Research Institute (TSRI) Assay Provider: Michael Jarstfer, University of North Carolina at Chapel Hill (UNC) Network: Molecular Libraries Probe Production Centers Network (MLPCN) Grant Proposal Number: 1 R03 MH085678-01A1 Grant Proposal PI: Michael Jarstfer, UNC External Assay ID: OTR_AG_FLUO8_1536_3XEC50 DRUN Name: Fluorescence-based cell-based high throughput dose response assay for agonists of the Oxytocin Receptor (OXTR). Description: Heterotrimeric G-protein coupled receptors (GPCRs) are major targets for disease therapeutics, due in part to their broad tissue distribution, structural diversity, varied modes of action, disease-associated mutations (1-4). Certain GPCRs are involved in signaling pathways for neuropeptides such as oxytocin, serotonin, and vasopressin, and influence complex social behaviors such as trust, anxiety, and parenting (5). Oxyto
- Fluorescence-based cell-based high throughput dose response assay for potentiators of Oxytocin Receptor (OXTR) Source (MLPCN Center Name): The Scripps Research Institute Molecular Screening Center (SRIMSC) Center Affiliation: The Scripps Research Institute (TSRI) Assay Provider: Michael Jarstfer, University of North Carolina at Chapel Hill (UNC) Network: Molecular Libraries Probe Production Centers Network (MLPCN) Grant Proposal Number: 1 R03 MH085678-01A1 Grant Proposal PI: Michael Jarstfer, UNC External Assay ID: OTR_POT_FLUO8_1536_3XEC50 DRUN Name: Fluorescence-based cell-based high throughput dose response assay for potentiators of Oxytocin Receptor (OXTR). Description: Heterotrimeric G-protein coupled receptors (GPCRs) are major targets for disease therapeutics, due in part to their broad tissue distribution, structural diversity, varied modes of action, disease-associated mutations (1-4). Certain GPCRs are involved in signaling pathways for neuropeptides such as oxytocin, serotonin, and vasopressin, and influence complex social behaviors such as trust, anxiety, and parenting (5). Oxyto
- Counterscreen for Oxytocin Receptor (OXTR) agonists: Fluorescence-based cell-based high throughput dose response assay to identify agonists of the vasopressin 1 receptor (V1R) Source (MLPCN Center Name): The Scripps Research Institute Molecular Screening Center (SRIMSC) Center Affiliation: The Scripps Research Institute (TSRI) Assay Provider: Michael Jarstfer, University of North Carolina at Chapel Hill (UNC) Network: Molecular Libraries Probe Production Centers Network (MLPCN) Grant Proposal Number: 1 R03 MH085678-01A1 Grant Proposal PI: Michael Jarstfer, UNC External Assay ID: V1R_AG_FLUO8_1536_3XEC50 OXTR DCSRUN Name: Counterscreen for Oxytocin Receptor (OXTR) agonists: Fluorescence-based cell-based high throughput dose response assay to identify agonists of the vasopressin 1 receptor (V1R). Description: Heterotrimeric G-protein coupled receptors (GPCRs) are major targets for disease therapeutics, due in part to their broad tissue distribution, structural diversity, varied modes of action, disease-associated mutations (1-4). Certain GPCRs are involved in signaling pathways for neuropeptides such as oxytocin, serotonin, and vasopressin, and influence c
- Counterscreen for vasopressin 1 receptor (V1R) agonists: Fluorescence-based cell-based high throughput dose response assay for agonists of the Oxytocin Receptor (OXTR) Source (MLPCN Center Name): The Scripps Research Institute Molecular Screening Center (SRIMSC) Center Affiliation: The Scripps Research Institute (TSRI) Assay Provider: Michael Jarstfer, University of North Carolina at Chapel Hill (UNC) Network: Molecular Libraries Probe Production Centers Network (MLPCN) Grant Proposal Number: 1 R03 MH085678-01A1 Grant Proposal PI: Michael Jarstfer, UNC External Assay ID: OTR_AG_FLUO8_1536_3XEC50 V1R DCSRUN Name: Counterscreen for vasopressin 1 receptor (V1R) agonists: Fluorescence-based cell-based high throughput dose response assay for agonists of the Oxytocin Receptor (OXTR). Description: Heterotrimeric G-protein coupled receptors (GPCRs) are major targets for disease therapeutics, due in part to their broad tissue distribution, structural diversity, varied modes of action, disease-associated mutations (1-4). Certain GPCRs are involved in signaling pathways for neuropeptides such as oxytocin, serotonin, and vasopressin, and influence complex so
- ChEMBL_1474014 (CHEMBL3419774) Competitive binding to SNAP-tagged oxytocin receptor (unknown origin) expressed in HEK293 cells incubated for 1 hr at RT followed by 4 hrs at 4 degC by TR-FRET assay in presence of 5-(3-(22-(3-carboxy-4-(3-oxo-3H-xanthen-9-yl)phenyl)-15,22-dioxo-2,5,8,11-tetraoxa-14,21-diazadocosyl)-5-(3-(2-chloro-4-fluorophenoxy)azetidin-1-yl)-4H-1,2,4-triazol-4-yl)-2-methoxypyridinium 2,2,2-trifluoroacetate
- ChEMBL_1474015 (CHEMBL3419775) Competitive binding to SNAP-tagged oxytocin receptor (unknown origin) expressed in HEK293 cells incubated for 1 hr at RT followed by 4 hrs at 4 degC by TR-FRET assay in presence of 2-(5-(3-(1-(5-(3-(2-chloro-4-fluorophenoxy)azetidin-1-yl)-4-(6-methoxypyridinium-3-yl)-4H-1,2,4-triazol-3-yl)-15-oxo-2,5,8,11-tetraoxa-14-azaoctadecan-18-yl)-1-ethyl-3-methyl-5-sulfoindolin-2-ylidene)penta-1,3-dienyl)-1-ethyl-3,3-dimethyl-3H-indolium-6-sulfonate 2,2,2-trifluoroacetate
- Fluorescence-based cell-based high throughput dose response screening assay for agonists of the vasopressin 1 receptor (V1R) Source (MLPCN Center Name): The Scripps Research Institute Molecular Screening Center (SRIMSC) Center Affiliation: The Scripps Research Institute (TSRI) Assay Provider: Michael Jarstfer, University of North Carolina at Chapel Hill (UNC) Network: Molecular Libraries Probe Production Centers Network (MLPCN) Grant Proposal Number: 1 R03 MH085678-01A1 Grant Proposal PI: Michael Jarstfer, UNC External Assay ID: V1R_AG_FLUO8_1536_3XEC50 DRUN Name: Fluorescence-based cell-based high throughput dose response screening assay for agonists of the vasopressin 1 receptor (V1R). Description: Heterotrimeric G-protein coupled receptors (GPCRs) are major targets for disease therapeutics, due in part to their broad tissue distribution, structural diversity, varied modes of action, disease-associated mutations (1-4). Certain GPCRs are involved in signaling pathways for neuropeptides such as oxytocin, serotonin, and vasopressin, and influence complex social behaviors such as trust, anxiety, and parent
 BDBM50205990 CHEMBL395429 OXYTOCIN
BDBM50205990 CHEMBL395429 OXYTOCIN BDBM50332723 Cystathionine Oxytocin CHEMBL1630531
BDBM50332723 Cystathionine Oxytocin CHEMBL1630531 CHEMBL352836 BDBM50056797 Oxytocin Trisulfide
CHEMBL352836 BDBM50056797 Oxytocin Trisulfide Lanthionine Oxytocin BDBM50332726 CHEMBL1630530
Lanthionine Oxytocin BDBM50332726 CHEMBL1630530 CAS_50-56-6 Oxytocin BDBM86210 NSC_439302
CAS_50-56-6 Oxytocin BDBM86210 NSC_439302 Cys-Tyr-Ile-Gln-Asn-Cys-Pro-Leu-Gly-NH2 1-[19-Amino-13-sec-butyl-10-(2-carbamoyl-ethyl)-7-carbamoylmethyl-16-(4-hydroxy-benzyl)-6,9,12,15,18-pentaoxo-1,2-dithia-5,8,11,14,17-pentaaza-cycloicosane-4-carbonyl]-pyrrolidine-2-carboxylic acid [1-(carbamoylmethyl-carbamoyl)-3-methyl-butyl]-amide CHEMBL265640 1-[19-Amino-13-sec-butyl-10-(2-carbamoyl-ethyl)-7-carbamoylmethyl-16-(4-hydroxy-benzyl)-6,9,12,15,18-pentaoxo-1,2-dithia-5,8,11,14,17-pentaaza-cycloicosane-4-carbonyl]-pyrrolidine-2-carboxylic acid [1-(carbamoylmethyl-carbamoyl)-3-methyl-butyl]-amide(Oxytocin) 1-[19-Amino-13-sec-butyl-10-(2-carbamoyl-ethyl)-7-carbamoylmethyl-16-(4-hydroxy-benzyl)-6,9,12,15,18-pentaoxo-1,2-dithia-5,8,11,14,17-pentaaza-cycloicosane-4-carbonyl]-pyrrolidine-2-carboxylic acid [1-(carbamoylmethyl-carbamoyl)-3-methyl-butyl]-amidec-[Cys-Tyr-Ile-Gln-Asn-Cys]-Pro-Leu-Gly-NH2 BDBM50013775 OXYTOCIN (oxytocin-OT) cyclo[Cys-Tyr-Ile-Gln-Asn-Cys]-Pro-Leu-Gly-NH2
Cys-Tyr-Ile-Gln-Asn-Cys-Pro-Leu-Gly-NH2 1-[19-Amino-13-sec-butyl-10-(2-carbamoyl-ethyl)-7-carbamoylmethyl-16-(4-hydroxy-benzyl)-6,9,12,15,18-pentaoxo-1,2-dithia-5,8,11,14,17-pentaaza-cycloicosane-4-carbonyl]-pyrrolidine-2-carboxylic acid [1-(carbamoylmethyl-carbamoyl)-3-methyl-butyl]-amide CHEMBL265640 1-[19-Amino-13-sec-butyl-10-(2-carbamoyl-ethyl)-7-carbamoylmethyl-16-(4-hydroxy-benzyl)-6,9,12,15,18-pentaoxo-1,2-dithia-5,8,11,14,17-pentaaza-cycloicosane-4-carbonyl]-pyrrolidine-2-carboxylic acid [1-(carbamoylmethyl-carbamoyl)-3-methyl-butyl]-amide(Oxytocin) 1-[19-Amino-13-sec-butyl-10-(2-carbamoyl-ethyl)-7-carbamoylmethyl-16-(4-hydroxy-benzyl)-6,9,12,15,18-pentaoxo-1,2-dithia-5,8,11,14,17-pentaaza-cycloicosane-4-carbonyl]-pyrrolidine-2-carboxylic acid [1-(carbamoylmethyl-carbamoyl)-3-methyl-butyl]-amidec-[Cys-Tyr-Ile-Gln-Asn-Cys]-Pro-Leu-Gly-NH2 BDBM50013775 OXYTOCIN (oxytocin-OT) cyclo[Cys-Tyr-Ile-Gln-Asn-Cys]-Pro-Leu-Gly-NH2